PROPIONIC ACID POTASSIUM SALT
potassium propionate
CAS: 327-62-8
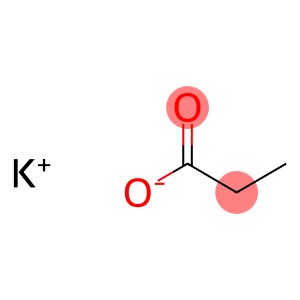
Molecular Formula: C3H5KO2
PROPIONIC ACID POTASSIUM SALT - Names and Identifiers
| Name | potassium propionate |
| Synonyms | Kaliumpropionat POTASSIUM PROPIONATE potassium propanoate potassium propionate Potassium propanoate Potassium propionate PROPIONIC ACID POTAS propanoicacid,potassiumsalt PROPIONIC ACID POTASSIUM SALT Propanoic acid, potassium salt Propanoic acid,potassiuM salt (9CI) |
| CAS | 327-62-8 |
| EINECS | 206-323-5 |
| InChI | InChI=1/C3H6O2.K/c1-2-3(4)5;/h2H2,1H3,(H,4,5);/q;+1/p-1 |
PROPIONIC ACID POTASSIUM SALT - Physico-chemical Properties
| Molecular Formula | C3H5KO2 |
| Molar Mass | 112.17 |
| Density | 1.438g/cm3 at 20℃ |
| Melting Point | 157 °C |
| Boling Point | 141.7°C at 760 mmHg |
| Flash Point | 57.7°C |
| Solubility | Methanol (Slightly, Heated), Water |
| Vapor Presure | 4.71hPa at 25℃ |
| Appearance | White crystal |
| Color | Off-White |
| Storage Condition | Inert atmosphere,Room Temperature |
| Stability | Hygroscopic |
| MDL | MFCD00045936 |
| Physical and Chemical Properties | White or colorless crystalline blocks. Soluble in water. Natural products are found in fermented foods, human sweat and ruminant digests. |
PROPIONIC ACID POTASSIUM SALT - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | 22 - Harmful if swallowed |
| Safety Description | S22 - Do not breathe dust. S24/25 - Avoid contact with skin and eyes. |
PROPIONIC ACID POTASSIUM SALT - Reference Information
| update date: | 2022/11/12 8:54:38 |
| LogP | -3.23 at 25℃ |
| introduction | potassium propionate, chemical formula KC3H5O2 · H2O. Molecular weight 130.19. White deliquescent crystals. The melting point is 410°C, which is the potassium salt of propionic acid. Easily soluble in water and ethanol. Heat to 120 ℃ and lose crystal water to become anhydrous salt. Natural potassium propionate is found in fermented foods, human sweat and ruminant digests. Industrial production is obtained by dissolving propionic acid in an aqueous solution of potassium hydroxide. |
| application | potassium propionate can be used as a food preservative. it has food label e code E283 in Europe and INS number 283 in Australia and new Zealand. |
| identification test | solubility soluble in water; Soluble in ethanol (OT-42). Potassium test (IT-27) positive propionic acid test positive sample and sulfuric acid co-temperature, should be propionic acid pungent smell. The positive organic acid, alkali and salt test will burn the sample at a lower temperature, and the remaining alkali will foam when exposed to acid. |
| content analysis | take 3g of sample (weighing to mg) that has been dried at 105 ℃ for 2h, put it into a distillation flask, and add 200ml of 50% phosphoric acid. Boil for 2h and collect the distillate. During the distillation process, water is added through the drip funnel to maintain the volume in the distillation bottle of about 200ml. Phenolphthalein test solution (TS-167) was used as indicator, and the distillate was titrated with lmol/L NaOH solution. Each Ml Imol/L NaOH solution is equivalent to 112.17mg of potassium propionate (C3 H5KO2). |
| usage limit | ADI is not restricted (FAO/WHO,2001). |
| uses | preservatives; Inhibitors of mold and bread microbial viscosity. EEC shall be used in dairy products, baked products, puddings and processed cheese. |
| production method | is obtained by neutralizing propionic acid and potassium hydroxide. |
Last Update:2024-04-09 15:16:49
Supplier List
Spot supply
Product Name: PROPIONIC ACID POTASSIUM SALT Visit Supplier Webpage Request for quotationCAS: 327-62-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Potassium Propionate Request for quotation
CAS: 327-62-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 327-62-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: Propionic Acid Potassium Salt Visit Supplier Webpage Request for quotationCAS: 327-62-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: PROPIONIC ACID POTASSIUM SALT Visit Supplier Webpage Request for quotationCAS: 327-62-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Potassium Propionate Request for quotation
CAS: 327-62-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 327-62-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: Propionic Acid Potassium Salt Visit Supplier Webpage Request for quotationCAS: 327-62-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


